Anresco tested a variety of cloth, medical, and N95 face masks to demonstrate their effectiveness at preventing the transmission of aerosol particles. Read more to learn about the results of our study.
Blog
By Zach Eisenberg Blog
Anresco studied the cannabinoid content of cannabis trimmings under different storage conditions over the period of a year to determine effects on degradation. Thermal rather than photodegradation was found to be the more significant pathway for reduced potential THC. Additionally, the most significant reductions in potential THC occurred in the first 30 days, regardless of storage conditions.
By Zach Eisenberg Blog
On July 2, 2020, the US Food and Drug Administration (FDA) issued a public warning regarding an increase in hand sanitizer products found to contain methanol but labeled as containing ethanol or isopropanol. Read more to learn about safety and efficacy issues with hand sanitizers currently on the market.
By Zach Eisenberg Blog
Terpenes are a group of organic compounds commonly found in plants and are primarily responsible for the aromatic characteristics of cannabis.
By Zach Eisenberg Blog
Read more to learn about Anresco's response to COVID-19.
By Zach Eisenberg Blog
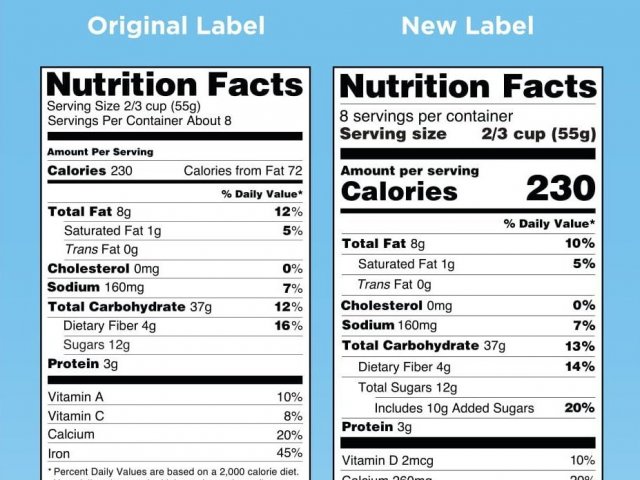
January 1, 2020 marks the compliance deadline of new FDA nutritional labeling requirements for companies with more than $10 million in annual food sales. While smaller companies have an additional year to comply, all imported foods must also meet these requirements.
By Zach Eisenberg Blog
Anresco analyzed a series of illicit market samples and found most were adulterated with high levels of vitamin e acetate — a compound frequently linked to the recent vaping crisis. Additionally, all samples failed the California BCC’s limits for pesticides, in some cases at hundreds of times the permitted level.
By Zach Eisenberg Blog
Anresco Labertories was awarded 'legacy business' status in San Francisco. Legacy Businesses are longstanding community-serving businesses that are recognized as valuable cultural assets to San Francisco by the Office of Small Business.
By Zach Eisenberg Blog
Click here to learn about the FDA's final rules to prevent intentional adulteration of food products.
By Zach Eisenberg Blog
Click here to learn about the nutrient database recently released by USDA/ARS.